
10.6 months, HR =0.56 95% CI, 0.46–0.69), despite 40.8% patients in the placebo group being cross-treated with pembrolizumab. Pembrolizumab plus pemetrexed-platinum resulted in a significant OS benefit over the placebo plus pemetrexed-platinum (22.0 vs. 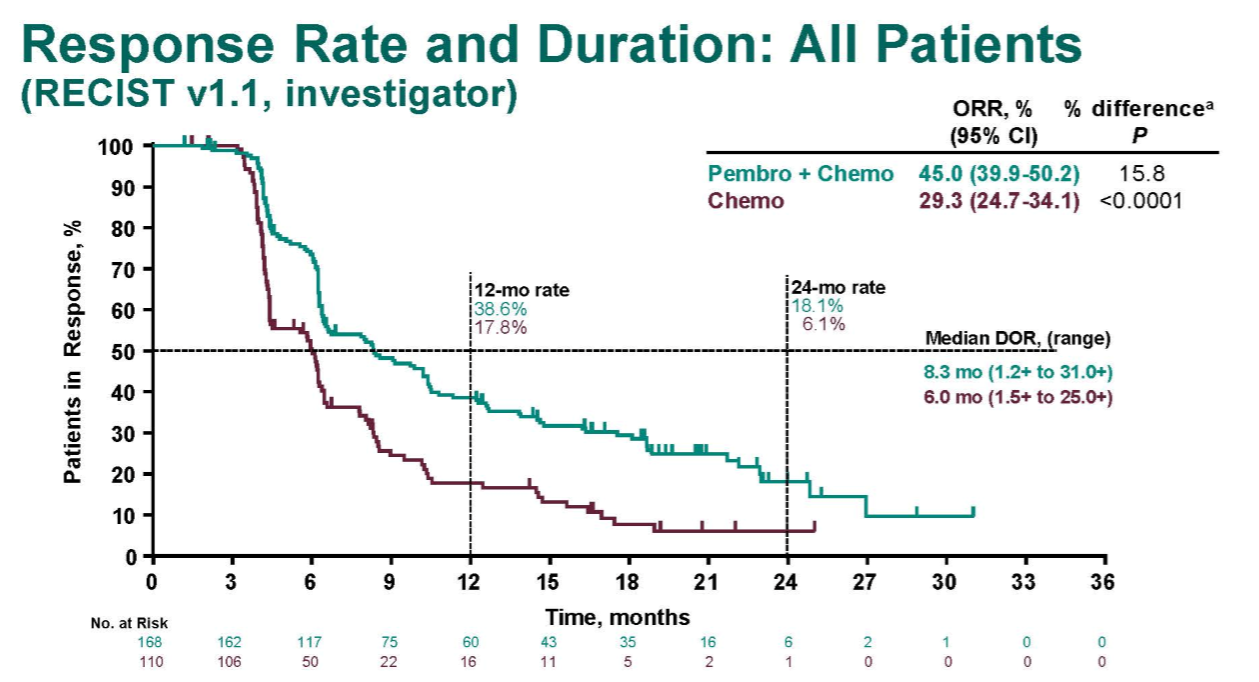
Addition of pembrolizumab to pemetrexed-platinum reduced the risk of disease progression by 51% versus standard of care chemotherapy (9.0 vs. According to the latest data released at the 2020 American Society of Clinical Oncology Annual Meeting ( 2), as of May 20, 2019, the median follow-up was 18.8 (range, 0.2–38.8) months. The primary end points were overall survival (OS) and progression-free survival (PFS). Crossover to pembrolizumab monotherapy was permitted among the patients in the placebo-combination group who had verified disease progression. Subsequent maintenance therapy with either pembrolizumab or placebo combined with pemetrexed was performed until the end of 35 cycles. Patients were randomly assigned in a 2:1 ratio to receive pemetrexed and a platinum-based drug plus either 200 mg of pembrolizumab or placebo every 3 weeks for 4 cycles. The KEYNOTE-189 study ( 1) is a randomized double-blind phase 3 trial with epidermal growth factor receptor (EGFR)- or anaplastic lymphoma kinase (ALK)-negative metastatic NSCLC patients who received no previous treatment. In particular, the lung cancer research in the KEYNOTE-189 trial has achieved excellent results. It binds to the programmed cell death protein 1 (PD-1) receptor and blocks its interaction with PD-L1 and PD-L2, which helps restore the immune response. Pembrolizumab (brand name Keytruda) is a humanized antibody which has been approved in many advanced cancers as a potent immunotherapy. Besides molecular targeted therapy, immunotherapies are also becoming potential treatment options for advanced NSCLC patients. With the progression of genomic medicine, precision oncology has contributed to improve survival outcomes and quality of life in NSCLC patients. Lung adenocarcinoma accounts for approximately 60% of all cases of non-small cell lung cancer (NSCLC).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
